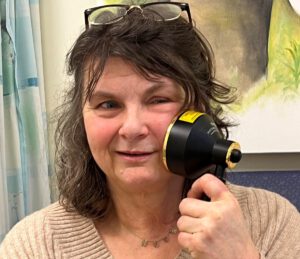 Cancer survivors are getting their lives back – thanks to a trail-blazing initiative at a bespoke post-cancer service at Nottingham City Hospital.
Cancer survivors are getting their lives back – thanks to a trail-blazing initiative at a bespoke post-cancer service at Nottingham City Hospital.
Nottingham Macmillan Late Effects helps people manage the physical, the psychological, and the psychosexual consequences of cancer treatment – and is the only place in the UK using Photobiomodulation (PBM) therapy to treat the long-term effects of radiotherapy.
Cindy Martin, 64, who was left with disfiguring scars after surgery for stage 4 cancer of the saliva gland, says PBM therapy has given her back her life.
“The radiotherapy was so intense, my skin became necrotic. By the time I found Late Effects, I was convinced I would always have open wounds on my face.”
Radiation-induced fibrosis – or tissue scarring – is completely unavoidable and can develop in the area being treated with radiotherapy years after the radiotherapy has stopped – with life-changing effects on patients.
“Looking through photographs from a year ago, my face was awful. Skin would come off in clumps, and I lost my earlobe. It affected my mobility – it was like I had a steel rod in my neck,” says Cindy.
Many patients are being cured of their cancer, but left with disabling and debilitating consequences.
“All my identity had been taken away,” says Cindy. “I couldn’t do my job any more, I’d lost loads of weight, and I looked different.”
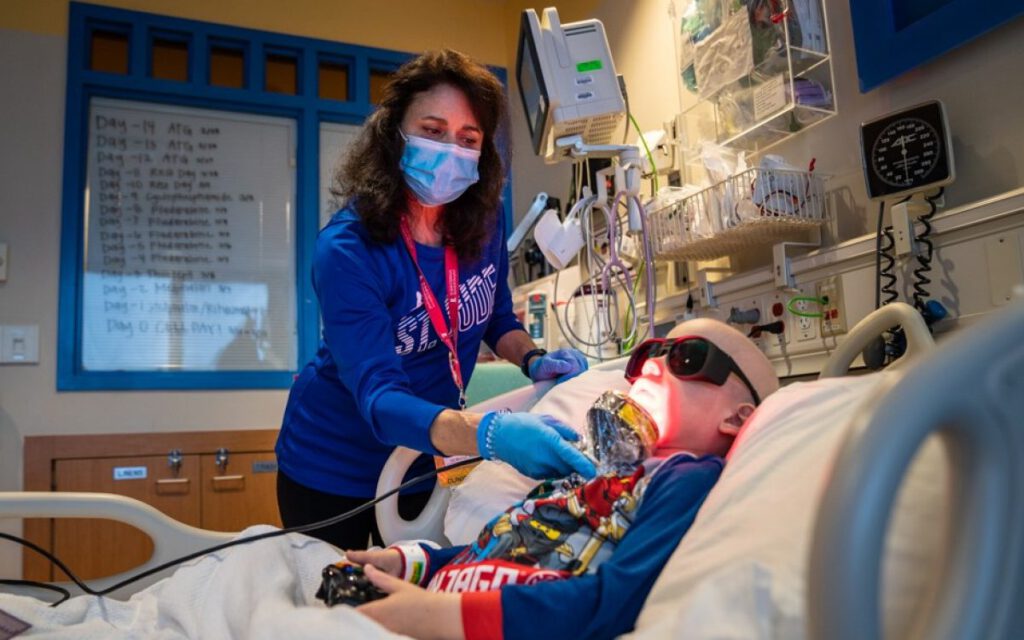
The PBM machine has a large ‘shower head’ attachment with LED lights. Patients are trained to administer the treatment themselves. The therapy is quick, simple, non-invasive, drug-free, and doesn’t cause any pain.
Emma Hallam is a Consultant Therapeutic Radiographer at Nottingham University Hospitals NHS Trust – and the person who thought of using PBM on radiation-induced fibrosis; it is usually used to treat lymphoedema – a long-term (chronic) condition that causes swelling.
Emma says: “We have people who have survivor’s guilt – they say that they feel lucky to be alive but sometimes wish that they hadn’t survived due to the long-term – sometimes disabling – side effects.
“Head and neck patients get much more radiation-induced fibrosis – they can’t eat, they can’t drink, can’t sleep – due to dry mouths and thick secretions.
“Our holistic approach helps improve quality of life and address all our patients’ needs, including the physical, psychological and psychosexual.”
PBM is just a small part of the Late Effects Service. The service uses a multi-disciplinary collaborative approach, with a dedicated rehabilitation and follow-up clinic, therapeutic radiographers, dieticians, and speech and language therapists.
“We are getting people swallowing again, who haven’t swallowed in years, people whose necks were immobile moving again, and people who haven’t been out of the house in years going out because they have got their lives back.”
Emma nominated colleague Kerry Clark for the The Skills for Health Our Heroes Clinical Support Worker of the Year award – and she is a finalist.
Kerry said: “It is such a humbling experience and a privilege to be able to offer help and support to patients who are feeling at such a loss after cancer treatment has ended.
“It is such a supportive environment to work in – I love my job and the difference we make to patients.”
And Cindy agrees. “Emma and her team have made such a difference to my life, I just can’t thank them enough – I wouldn’t be here today if it wasn’t for them. I was alive but I wasn’t living.”
The Skills for Health Our Health Heroes Awards is sponsored by NHS England, NHS Employers, NHS Shared Business Services and more. Gold, silver and bronze Clinical Support Worker of the Year award winners will be announced at a ceremony in London on 16 April. Show your support for Kerry by casting your vote here:
https://www.skillsforhealth.org.uk/awards/
Read more about the Nottingham Macmillan Late Effects Service.
Notes
Emma now leads on the early management of all head and neck lymphedema, under the expert guidance of the specialist lymphedema team.
Late Effects has developed patient questionnaires as a 10-year monitoring system; identifying late effects early is important as fibrosis becomes more resistant to therapy as it builds up.
Peer-to-peer knowledge sharing across the world, and working with patients and UK charities has been key to educating people of the need for late effects services; Nottingham has been pivotal in helping establish around 20 similar services across the UK, and helped create best practice guidelines with the Pelvic Radiation Disease Association. Emma and her team hope to work with head and neck cancer charities and other late effects services to produce best practice guidelines for this patient group.
Article Source: https://www.nuh.nhs.uk
 Featured Testimonials
Featured Testimonials



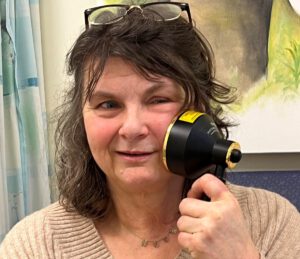









 The paper “High-intensity versus low-level laser therapy in the treatment of patients with knee osteoarthritis: a randomized controlled trial” Kheshie et al 2014 pitches the 3B BTL laser against the HIRO class IV “High intensity” laser. The results appear to show that the HIRO class IV “High intensity laser” was more effective than the “Low Level Laser”.
The paper “High-intensity versus low-level laser therapy in the treatment of patients with knee osteoarthritis: a randomized controlled trial” Kheshie et al 2014 pitches the 3B BTL laser against the HIRO class IV “High intensity” laser. The results appear to show that the HIRO class IV “High intensity laser” was more effective than the “Low Level Laser”.