Categories
Essays
Links
Category Archives: Industry
Children’s Health Ireland: Photobiomodulation for the treatment of cancer therapy side effects
THOR is helping to improve the quality of life of Children’s Health Ireland patients who are undergoing treatment for cancer, and will not rest until every single patient in need has access to our Photomedicine Photobiomodulation (red light) therapy.
Posted in Industry, Media/News Videos, PBM/LLLT, Video of the Week
on Children’s Health Ireland: Photobiomodulation for the treatment of cancer therapy side effects
Leeds Teaching Hospitals NHS Trust: Mucositis and Photobiomodulation video
Oral mucositis is a term used to describe having a sore mouth, often with multiple painful ulcers. It is a side effect of cancer treatment that affects up to eight in every ten children who have chemotherapy.
This is a film by Leeds Teaching Hospitals NHS Trust about mucositis and a light treatment (called Photobiomodulation) that can be used to prevent and treat this. It has been designed and filmed with children and young people who have had mucositis, and those who have had the light treatment.
This video was made as part of a research project, that aimed to find out how acceptable the light treatment was to children as part of their cancer treatment. The children and young people involved in this video hope that it will help to tell other children and their families what mucositis can be like, and what to expect with the light treatment.
You can watch the video on lchtv.com/photobiomodulation
For more, please visit Leeds Hospitals Charity and Leeds Teaching Hospitals NHS Trust
More Videos | NovoTHOR Testimonials | NovoTHOR Treatment Locations
View current photobiomodulation therapy training dates in your location.
Posted in Industry, Testimonials, Video of the Week
on Leeds Teaching Hospitals NHS Trust: Mucositis and Photobiomodulation video
Making the hardest days easier – photobiomodulation for treatment of oral mucositis
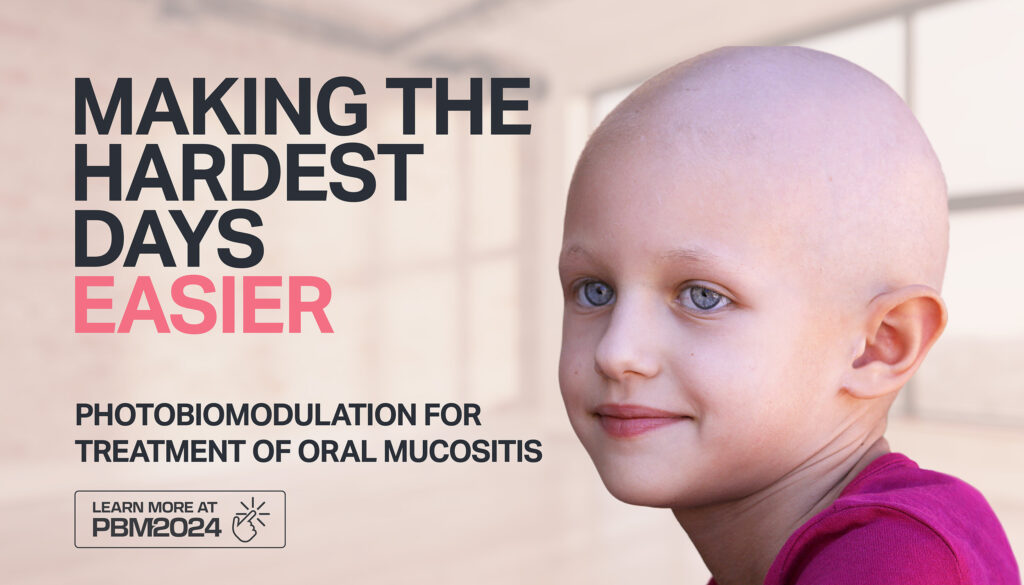
Oral mucositis is a common side effect of high-dose chemotherapy or radiotherapy, leaving many patients unable to eat, drink, or swallow and consequently requiring tube feeding.
Until recently, there had been no satisfactory treatment or method of prevention. However, Photobiomodulation (PBM), a light therapy treatment proven safe and effective by the National Institute for Health and Care Excellence (NICE), is now widely used across the NHS with incredible results.
Jenny Gale, a head and neck clinical nurse specialist at East Suffolk and North Essex NHS Trust, said: “It can really make your mouth sore, and it hurts to swallow, which can make eating and drinking difficult, as well as impacting your general wellbeing.”
She added: “PBM is completely painless and very simple to carry out; most patients can do it themselves.
“Evidence shows patients need less pain relief when they’ve had PBM, and they generally cope with their treatment better. It’s really exciting that we are offering it in our hospitals now.”
The breakthrough allowing NHS adoption of PBM is due to the development of the THOR LED lollipop, which patients use by placing it in their mouth for 60 seconds on the tongue and 60 seconds under the tongue.
Photobiomodulation is a low-intensity light therapy that stimulates mitochondria to produce more energy (ATP) and reduce inflammation, rapidly reducing pain and improving healing rates.
It can be used prophylactically to prevent the incidence and severity of oral mucositis or therapeutically after mucositis has developed.
Both the Christie and Leeds Cancer Centres found that PBM reduced morphine prescriptions by 63 per cent and 45 per cent, respectively, in their audits and reduced hospital bed days by 88.7 per cent at Christie and hospitalisation by 89 per cent at Leeds.
There is a rare opportunity to attend the World Association for Photobiomodulation conference, which will be held in London this year from August 23 to 25, where many NHS users of PBM will be presenting their data and discussing it one-on-one with delegates.
Visit PBM2024.com for details.
Posted in Industry, PBM/LLLT, THE FUTURE OF PBM/LLLT
on Making the hardest days easier – photobiomodulation for treatment of oral mucositis
I was alive, but now I’m living again, says cancer survivor Cindy
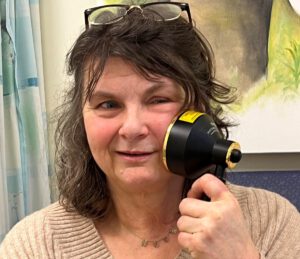 Cancer survivors are getting their lives back – thanks to a trail-blazing initiative at a bespoke post-cancer service at Nottingham City Hospital.
Cancer survivors are getting their lives back – thanks to a trail-blazing initiative at a bespoke post-cancer service at Nottingham City Hospital.
Nottingham Macmillan Late Effects helps people manage the physical, the psychological, and the psychosexual consequences of cancer treatment – and is the only place in the UK using Photobiomodulation (PBM) therapy to treat the long-term effects of radiotherapy.
Cindy Martin, 64, who was left with disfiguring scars after surgery for stage 4 cancer of the saliva gland, says PBM therapy has given her back her life.
“The radiotherapy was so intense, my skin became necrotic. By the time I found Late Effects, I was convinced I would always have open wounds on my face.”
Radiation-induced fibrosis – or tissue scarring – is completely unavoidable and can develop in the area being treated with radiotherapy years after the radiotherapy has stopped – with life-changing effects on patients.
“Looking through photographs from a year ago, my face was awful. Skin would come off in clumps, and I lost my earlobe. It affected my mobility – it was like I had a steel rod in my neck,” says Cindy.
Many patients are being cured of their cancer, but left with disabling and debilitating consequences.
“All my identity had been taken away,” says Cindy. “I couldn’t do my job any more, I’d lost loads of weight, and I looked different.”
The PBM machine has a large ‘shower head’ attachment with LED lights. Patients are trained to administer the treatment themselves. The therapy is quick, simple, non-invasive, drug-free, and doesn’t cause any pain.
Emma Hallam is a Consultant Therapeutic Radiographer at Nottingham University Hospitals NHS Trust – and the person who thought of using PBM on radiation-induced fibrosis; it is usually used to treat lymphoedema – a long-term (chronic) condition that causes swelling.
Emma says: “We have people who have survivor’s guilt – they say that they feel lucky to be alive but sometimes wish that they hadn’t survived due to the long-term – sometimes disabling – side effects.
“Head and neck patients get much more radiation-induced fibrosis – they can’t eat, they can’t drink, can’t sleep – due to dry mouths and thick secretions.
“Our holistic approach helps improve quality of life and address all our patients’ needs, including the physical, psychological and psychosexual.”
PBM is just a small part of the Late Effects Service. The service uses a multi-disciplinary collaborative approach, with a dedicated rehabilitation and follow-up clinic, therapeutic radiographers, dieticians, and speech and language therapists.
“We are getting people swallowing again, who haven’t swallowed in years, people whose necks were immobile moving again, and people who haven’t been out of the house in years going out because they have got their lives back.”
Emma nominated colleague Kerry Clark for the The Skills for Health Our Heroes Clinical Support Worker of the Year award – and she is a finalist.
Kerry said: “It is such a humbling experience and a privilege to be able to offer help and support to patients who are feeling at such a loss after cancer treatment has ended.
“It is such a supportive environment to work in – I love my job and the difference we make to patients.”
And Cindy agrees. “Emma and her team have made such a difference to my life, I just can’t thank them enough – I wouldn’t be here today if it wasn’t for them. I was alive but I wasn’t living.”
The Skills for Health Our Health Heroes Awards is sponsored by NHS England, NHS Employers, NHS Shared Business Services and more. Gold, silver and bronze Clinical Support Worker of the Year award winners will be announced at a ceremony in London on 16 April. Show your support for Kerry by casting your vote here:
https://www.skillsforhealth.org.uk/awards/
Read more about the Nottingham Macmillan Late Effects Service.
Notes
Emma now leads on the early management of all head and neck lymphedema, under the expert guidance of the specialist lymphedema team.
Late Effects has developed patient questionnaires as a 10-year monitoring system; identifying late effects early is important as fibrosis becomes more resistant to therapy as it builds up.
Peer-to-peer knowledge sharing across the world, and working with patients and UK charities has been key to educating people of the need for late effects services; Nottingham has been pivotal in helping establish around 20 similar services across the UK, and helped create best practice guidelines with the Pelvic Radiation Disease Association. Emma and her team hope to work with head and neck cancer charities and other late effects services to produce best practice guidelines for this patient group.
Article Source: https://www.nuh.nhs.uk
Posted in Industry, Testimonials, THE FUTURE OF PBM/LLLT
on I was alive, but now I’m living again, says cancer survivor Cindy
THOR and PBM making national news
The BBC and Daily Mail are helping to reveal PBM (Photobiomodulation Therapy) into the wider community. With more public awareness of the amazing benefits how PBM light therapy is massively improving lives, we hope it will encourage more people to search it out and ask for this treatment in their own locations.
Read the BBC article:
https://www.bbc.com/news/articles/cg3ee8xl96eo
Read the Daily Mail PDF article:
Daily Mail Gadget that shines bright red LEDs to mend cancer scars now being used to treat debilitating scarring and swelling in the mouth caused by radiotherapy
Posted in Industry, PBM/LLLT, THE FUTURE OF PBM/LLLT
on THOR and PBM making national news
New light therapy helps to ease pain for head and neck cancer patients
Cancer patients are benefiting from a new light therapy treatment which tackles a common side effect caused by chemotherapy and radiotherapy.
Photobiomodulation (PBM) has launched in hospitals run by East Suffolk and North Essex NHS Foundation Trust (ESNEFT) to prevent or treat oral mucositis. It occurs when cancer treatments damage the lining of the mouth. This can leave the tissue vulnerable to ulceration and infection.
All head and neck cancer patients having radiotherapy are being offered PBM at Colchester and Ipswich hospitals.
The treatments are being overseen by Jenny Gale, radiotherapy sister at Colchester, and Lyndsey Rew Macmillan specialist radiographer at Ipswich.
Jenny is also interim head and neck clinical nurse specialist at ESNEFT. She has had radiotherapy treatment herself so is able to relate to what her patients are feeling and the benefits PBM will bring for them.
She said: “It can really make your mouth sore. It hurts to swallow too which can make eating and drinking difficult, as well as having an impact on your general wellbeing.
“Unlike the side effects, PBM is completely painless. It’s very simple to carry out so most patients can do it themselves and take control of their treatment which is amazing.
“Evidence shows patients need less pain relief when they’ve had PBM, and they generally cope with their treatment better. It’s really exciting that we are offering it in our hospitals now.”
PBM works by applying infrared light to tissue, via two probes, which can reduce inflammation and improve healing.
Funding for two Thor laser machines, one at Colchester Hospital and the other at Ipswich Hospital, was provided thanks to a £31,000 grant from Colchester & Ipswich Hospitals Charity.
To read the full article, go here:
oncologynewstoday.co.uk
Posted in Industry, PBM/LLLT, THE FUTURE OF PBM/LLLT
on New light therapy helps to ease pain for head and neck cancer patients
Pope Francis praises Light Therapy after recovery
 Pope Francis credits Light Therapy for his recovery from a wheelchair during a ceremony on Saturday, April 29, during his homily at St. Elisabeth Church in Budapest. Pope Francis spoke of how the red-light therapy healed his fractured knee. The Pope was able to stand up out of his wheelchair and walk with a and walk with a cane.
Pope Francis credits Light Therapy for his recovery from a wheelchair during a ceremony on Saturday, April 29, during his homily at St. Elisabeth Church in Budapest. Pope Francis spoke of how the red-light therapy healed his fractured knee. The Pope was able to stand up out of his wheelchair and walk with a and walk with a cane.
To read the full article, go here: https://themissionstribune.com/headlines
Posted in Industry, Special Feature
on Pope Francis praises Light Therapy after recovery
 Featured Testimonials
Featured Testimonials

