Categories
Essays
Links
Author Archives: James Carroll
Emma Hallam Interview – Treatment of late effects of cancer treatment with PBM

This is a compelling interview with Emma Hallam, Macmillan Consultant Radiographer in Post Treatment & Late Effects at Nottingham Radiotherapy Centre.
The side effects and late effects (delayed effects) of cancer treatment can be many (see the list below), painful, unsightly, life long and life-limiting. Patients are frequently unprepared, and medical solutions have been inadequate.
 Emma has been using PBM for the last three years (since 2021) at Nottingham University Hospitals NHS Trust, one of the UK’s largest and busiest teaching hospitals. She has collected data on over 150 patients comparing baseline data on pain, function and quality of life with results after twice-weekly treatments for 6 weeks or weekly treatments for 12 weeks. She is particularly convinced and excited by the benefits of THOR PBM on radiation-induced fibrosis (RIF), which is a major contribution to many of the late effects listed below.
Emma has been using PBM for the last three years (since 2021) at Nottingham University Hospitals NHS Trust, one of the UK’s largest and busiest teaching hospitals. She has collected data on over 150 patients comparing baseline data on pain, function and quality of life with results after twice-weekly treatments for 6 weeks or weekly treatments for 12 weeks. She is particularly convinced and excited by the benefits of THOR PBM on radiation-induced fibrosis (RIF), which is a major contribution to many of the late effects listed below.
Emma’s amazing work has LED to the creation of 25 late-effects clinics across the UK, and she is only just getting started. Emma’s dedication to patients and results with PBM have been remarkable, and listening to her story is compelling.
Click to hear the interview
https://www.buzzsprout.com/2314183/15767123
OR LISTEN ON APPLE PODCASTS
OR LISTEN ON SPOTIFY
Click to contact a lovely THOR Oncology PBM product specialist
https://www.thorlaser.com/purchase-enquiry.php
Table of diseases
| Pelvis | Breast | Head & Neck |
| Pelvic Radiation Disease Gastrointestinal Urological Gynaecological Sexual function Bone Lumbosacral plexopathy Peripheral neuropathy Fistulas Skin changes Necrotic ulcers Lymphoedema Menopausal symptoms |
Hardening & shrinkage Skin changes Lymphoedema Arm & shoulder dysfunction Peripheral neuropathy Nerve damage Lung changes Breathlessness Menopausal symptoms Morphea Peripheral neuropathy of the diaphragm Cardiac issues |
Xerostomia Taste changes Thick secretions Dysphagia Weight loss & malnutrition Dental decay Trismus Skin changes Neck & shoulder dysfunction Lymphoedema Peripheral neuropathy Tinnitus & hearing loss Balance issues Thyroid issues Osteoradionecrosis (ORN) Burning tongue syndrome Dropped head syndrome |
Thank you, Emma.
Listen here: Support the show
Posted in Research
on Emma Hallam Interview – Treatment of late effects of cancer treatment with PBM
Dr Ben from Health Versus Wealth interviews THOR CEO James Carroll
This is an interview featuring me by Dr. Ben Sinclair, a British physician / general practitioner (GP) Sinclair. His podcast is called Health Versus Wealth. Primarily, this podcast is about my health choices and routines. He describes me as a “highly driven entrepreneur, 61 years old (now 62), of THOR Laser company (correction: THOR Photomedicine Ltd and THOR Photomedicine Inc), of which he is is Founder CEO, which specialises in design and manufacture of Photobiomodulation (PBM) devices. THOR £10 million a year in sales, has 5,000 customers in 70 countries, and is marking THOR’s 30th anniversary.” Dr Ben says I am “a businessman who prioritises his wealth, in that he works 364 days a year! (actually, my motivation is establishing PBM as a first-line medical treatment in every department of every hospital in the whole world), wealth might be an outcome, but it is not the goal.) How does this impact his work-life balance? “The only day I take off is Christmas Day,” he says. Despite this, he says he’s in fine health, fit and eats a nutritious diet.
THOR’s celebrity clients include Mo Farah (Mo only used NovoTHOR whilst training with the Nike, Oregon project), Novak Djokovic, Hollywood actor Mark Wahlberg, and his biggest-name customer, film star Tom Cruise. Cruise famously broke his ankle during filming in London on ‘Mission Impossible: Fallout’ in 2017 while leaping from one building to another. Cruise used James’s laser to recover, and now he has lasers in his homes and is always one on location. James LED a workshop in Florida, and Cruise walked in wearing his trademark aviator sunglasses. “Tom was a delight,” says James. As for his business ambitions to grow the company, James adds: “My real goal is to get this technology into every corner of every department of every hospital in the whole world.”
Posted in Interviews, James in Action, THE FUTURE OF PBM/LLLT
on Dr Ben from Health Versus Wealth interviews THOR CEO James Carroll
The evidence for Photobiomodulation treatment of musculoskeletal pain
THERAPY EXPO SYNOPSIS: Photobiomodulation (PBM) is one of the best researched physical therapy modalities. More than 800 RCTs have been published on the benefits of PBM, thousands of papers published on the mechanism of action and dose response, more than 8,000 academic papers in total. Systematic reviews and clinical guidelines recommending PBM have been published in the worlds leading journals including The Lancet, British Journal of Sports Medicine, the British Medical Journal and JAMA for a wide range of musculoskeletal disorders. The American College of Physicians recommend PBM for Low Back Pain, and NICE recommend PBM for Oral Mucositis. PBM works primarily on the powerhouse of the cell (mitochondria), increasing energy (ATP) and reducing oxidative stress (cytokines) that lead to inflammation cell death and diseases associated with ageing. The unique feature of PBM is its ability to switch on tissue and regenerative processes. This presentation summarises the evidence for PBM in musculoskeletal pain.
For more on PBM evidence, training or products just click
Posted in Research
on The evidence for Photobiomodulation treatment of musculoskeletal pain
James Carroll 2020 WALT Presidential Commendation
Thank you to The World Association for Photobiomodulation Therapy for the award today. As I said in my acceptance speech I am grateful for the many friends and good times I have enjoyed in the field of LLLT/PBM over the last 33 years, largely thanks to WALT. I should have said more about what I have learned from the many scientists, doctors, dentists, veterinarians, therapists, nurses and patients. It has LED to an exciting, rewarding and fulfilling career, and I look forward to the next 40 years as we take PBM to where it is needed and belongs. So thank you WALT for giving me a life worth living.
Posted in James in Action, Special Feature
on James Carroll 2020 WALT Presidential Commendation
RIP Dr Kevin Moore who died last week 15 July 2020
 Kevin was a husband, father, anaesthetist, chronic pain specialist, researcher, a fun, kind and generous man.
Kevin was a husband, father, anaesthetist, chronic pain specialist, researcher, a fun, kind and generous man.
He was a vital founding member of the World Association for Laser Therapy (WALT) and a long-standing cheerleader for Photobiomodulation (PBM) as it is now known.
As well as being Consultant Anaesthetist at The Royal Oldham Hospital UK, Kevin was medical director at Dr Kershaw’s Hospice.
Kevin also had the thankless task of being the WALT treasurer for many years. His leadership and steady hand ensured the organisation survived several problematic periods.
All who met him will remember his warmth, humour and generosity of spirit.
He will be forever memorialised in my LLLT/PBM presentations as he researched and published two of my favourite PBM papers of all time (abstracts below and links to some PDFs).
I have converted some of his slides into this 4 minute movie and managed to lay some audio over it which was recorded about 20 years ago. Given this is one of the worst kinds of pain any one can suffer it is remarkable that such a simple tool can achieve such good results.
So thank you to Kevin for your contribution to the world, and our condolences to Jill, his wife and his children. Kevin was a special man, I know he will be missed.
His local new paper reports his death here https://www.oldham-chronicle.co.uk/news-features/139/main-news/135491/tributes-paid-to-muchloved-dr-moore
Other newspaper reports about Kevin https://www.manchestereveningnews.co.uk/news/local-news/easing-the-burden-for-patients-in-pain-1134114
ABSTRACTS WITH PDF LINKS BELOW
THE EFFECT OF INFRARED LASER IRRADIATION (LLLI) ON THE DURATION AND SEVERITY OF POSTOPERATIVE PAIN: A DOUBLE BLIND TRIAL
Kevin C. Moore, Naru Hira, Ian J. Broome* and John A. Cruikshank
Departments of Anaesthesia and General Surgery, The Royal Oldham Hospital, Oldham, U.K *Department of Anaesthesia, The Royal Hallamshire Hospital, Sheffield, U.K., General Practitioner, Pennymeadow Clinic, Ashton-under-Lyne, U.K.
This trial was designed to test the hypothesis that LLLT reduces the extent and duration of postoperative pain. Twenty consecutive patients for elective cholecystectomy were randomly allocated for either LLLT or as controls. The trial was double blind. Patients for LLLT received 6- 8-min treatment (GaAlAs: 830 nm: 60 mW CW: CM) to the wound area immediately following skin closure prior to emergence from GA. All patients were prescribed on demand postoperative analgesia (IM or oral according to pain severity). Recordings of pain scores (0-10) and analgesic requirements were noted by an independent assessor. There was a significant difference in the number of doses of narcotic analgesic (IM) required between the two groups. Controls n = 5.5: LLLT n = 2.5. No patient in the LLLT group required IM analgesia after 24 h. Similarly the requirement for oral analgesia was reduced in the LLLT group. Controls n = 9: LLLT n = 4. Control patients assessed their overall pain as moderate to severe compared with mild to moderate in the LLLT group. The results justify further evaluation on a larger trial population
Laser Therapy, 4: 145-150. 1992
A DOUBLE BLIND CROSSOVER TRIAL OF LOW LEVEL LASER THERAPY IN THE TREATMENT OF POSTHERPETIC NEURALGIA
Moore, K.C., Hira, N., Kumar, P.S., Jayakumar, C.S., and Ohshiro, T
Postherpetic. neuralgia can be an extremely painful condition which in many cases proves resistant lo all the accepted forms of treatment. It is frequently most severe in the elderly and may persist for years with no predictable course.
This trial was designed as a double blind assessment of the efficacy of low level laser therapy in the relief of the pain of post herpetic neuralgia with patients acting as their own controls. Admission to the trial was limited to patients with established post herpetic neuralgia of at least six months duration and who had shown little or no response to conventional methods of treatment. Measurements of pain intensity and distribution were noted over a period of eight treatments in two groups of patients each of which received four consecutive laser treatments. The results demonstrate a significant reduction in the pain intensity and distribution following a course of low level laser therapy.
Laser Therapy Pilot Edition 1:0 Pilot Issue 2 Pages 61-64
Posted in Research
on RIP Dr Kevin Moore who died last week 15 July 2020
LumiThera Enrolls First Patient in the U.S. Multi-Center, LIGHTSITE III Clinical Study to Treat Dry Age-Related Macular Degeneration
SEATTLE, Oct. 1, 2019 /PRNewswire/ — LumiThera Inc., a commercial stage medical device company delivering Photobiomodulation (PBM) treatment for ocular disorders and disease, today announced it has begun enrolling patients in a multi-center United States clinical study in dry Age-Related Macular Degeneration (AMD) patients.
The randomized, multi-center study called LIGHTSITE III enrolled and treated the first patient at Cumberland Valley Retina in Hagerstown, Maryland. The United States study is being conducted in leading retinal centers throughout the United States. The study will enroll approximately 100 patients suffering from dry AMD and treat them over the course of two years. In addition to demonstrating safety, key efficacy endpoints include visual acuity, contrast sensitivity and reduction of drusen deposits.
In February, the company announced that the National Institute of Health and the division of the National Eye Institute was providing a $2.5M grant to partially support the U.S. study.
“This is an exciting opportunity to establish a potential new treatment for dry AMD patients” remarked Allen Hu, M.D. (Principal Investigator, Cumberland Valley Retina Consultants). “The previous work with PBM has been very promising. Our center has been involved in many drug studies with the wet form of AMD, but this study critically provides an important medical device treatment for the majority of patients with intermediate dry AMD and limited options.”
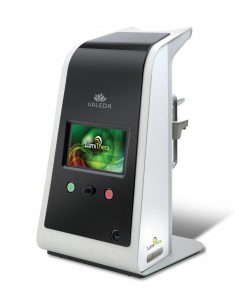
LumiThera has already obtained a CE mark authorization to commercialize the Valeda™ Light Delivery System in the European Economic Area for the treatment of dry AMD. The separate LIGHTSITE II study is an ongoing post-marketing study with top retinal centers in key European countries and was initiated earlier in the year.
“The LIGHTSITE III study has engaged eight top U.S. retinal centers to evaluate the Valeda,” stated Eleonora Lad, M.D., Ph.D. (Principal Investigator, Duke University). “We are now actively screening patients and will be measuring multiple vision outcome measures. The Duke Imaging Center with Dr. Glenn Jaffe will also be evaluating retina images for all study centers involved to determine if we are improving the health of the eye during the course of the trial. This is a major study that will establish a novel treatment for these elderly patients and could have a big impact on their quality of life.”
“We have two multi-center trials currently underway, one in Europe and one in the U.S.,” stated Clark Tedford, Ph.D., President and CEO. The data from these trials will be used to further support our commercialization efforts and global regulatory submissions. We have enlisted top private retinal and University centers in the U.S. for the LIGHTSITE III trial and if successful, the study would allow an important treatment choice in the prevention of vision loss for patients here.”
About LumiThera Inc.
LumiThera is a commercial-stage medical device company focused on treating people affected by ocular disorders and diseases including dry AMD, a leading cause of blindness in adults over 65. The company is a leader in the use of PBM for treatment of acute and chronic ocular diseases and disorders. The company is developing the office-based Valeda Light Delivery System to be used by eye care specialists as medical treatments.
The Valeda Light Delivery System has been granted authorization to use the CE Mark by an EU Notified Body as required for commercial use in the European Economic Area only. Valeda is not approved for use by the Food & Drug Administration (FDA) in the USA.
View the press release on Lumithera website at www.lumithera.com
Disclosure: I am an investor in Lumithera a) because it successfully treats an unmet medical need, (dry AMD) b) I think it will be the first billion dollar photobiomodulation company.
Posted in Industry
on LumiThera Enrolls First Patient in the U.S. Multi-Center, LIGHTSITE III Clinical Study to Treat Dry Age-Related Macular Degeneration
Valeda Light Delivery System Receives 2019 Medical Design Excellence Award
SEATTLE, June 12, 2019 /PRNewswire/ — LumiThera, Inc. and Product Creation Studio today announced that LumiThera’s Valeda Light Delivery System has been honored with a silver award in the 21st Annual Medical Design Excellence Awards competition. The 2019 winners were announced at the MDEA Ceremony on Tuesday, June 11, 2019 in conjunction with the MD&M East event in New York, New York.
LumiThera, Inc. is a leader in the use of Photobiomodulation for the treatment of acute and chronic ocular diseases and disorders. Age-related macular degeneration (AMD) is the leading cause of blindness in adults over 65. Product Creation Studio worked with LumiThera from product concept through transfer to manufacturing to design and engineer a medical device that would accurately, safely, and effectively treat patients suffering from dry AMD. The resulting Valeda Light Delivery System is currently the only approved treatment using Photobiomodulation for dry age-related macular degeneration in the European Union.
The MDEA are the medtech industry’s premier design competition committed to recognizing significant achievements in medical product design and engineering that improve the quality of healthcare delivery and accessibility. The awards program celebrates the accomplishments of the medical device manufacturers, their suppliers, and the many people behind the scenes—engineers, scientists, designers, and clinicians—who are responsible for the cutting-edge products that are saving lives; improving patient healthcare; and transforming medtech—one innovation at a time.
“This award recognizes the efforts of the LumiThera and the Product Creation teams who have worked diligently to deliver a life changing treatment for those suffering from dry AMD,” said Clark Tedford, PhD, CEO and President. “We are pleased to bring the Valeda Light Delivery System to the ophthalmic market and offer dry AMD patients a treatment for this debilitating disease.”
“Product Creation Studio’s collaboration with LumiThera embodies what our organization is about: empowering innovators with the right design and development services at the right time to deliver products with impact,” Scott Thielman, Chief Technology officer of Product Creation Studio, said. “The Valeda Light Delivery System is a breakthrough product that makes a meaningful difference for patients and their families. It has been an honor to support the LumiThera team with comprehensive engineering and design services from the company’s early days through production.”
The 2019 MDEA Juror Panel selected 44 exceptional finalists in nine medical technology product categories. Products were judged based on design and engineering innovation; function and user-related innovation; patient benefits; business benefits; and overall benefit to the healthcare system. Unlike other design competitions that are merely styling contests, the MDEA jury is comprised of a balance of practicing doctors, nurses, and technicians alongside industrial designers, engineers, manufacturers, and human factors experts.
View the press release on Lumithera website at www.lumithera.com
Disclosure: I am an investor in Lumithera a) because it successfully treats an unmet medical need, (dry AMD) b) I think it will be the first billion dollar photobiomodulation company.
Posted in Industry
on Valeda Light Delivery System Receives 2019 Medical Design Excellence Award
 Featured Testimonials
Featured Testimonials