Author Archives: James Carroll
Emma Hallam Interview – Treatment of late effects of cancer treatment with PBM

This is a compelling interview with Emma Hallam, Macmillan Consultant Radiographer in Post Treatment & Late Effects at Nottingham Radiotherapy Centre.
The side effects and late effects (delayed effects) of cancer treatment can be many (see the list below), painful, unsightly, life long and life-limiting. Patients are frequently unprepared, and medical solutions have been inadequate.
 Emma has been using PBM for the last three years (since 2021) at Nottingham University Hospitals NHS Trust, one of the UK’s largest and busiest teaching hospitals. She has collected data on over 150 patients comparing baseline data on pain, function and quality of life with results after twice-weekly treatments for 6 weeks or weekly treatments for 12 weeks. She is particularly convinced … Continue reading
Emma has been using PBM for the last three years (since 2021) at Nottingham University Hospitals NHS Trust, one of the UK’s largest and busiest teaching hospitals. She has collected data on over 150 patients comparing baseline data on pain, function and quality of life with results after twice-weekly treatments for 6 weeks or weekly treatments for 12 weeks. She is particularly convinced … Continue reading
Dr Ben from Health Versus Wealth interviews THOR CEO James Carroll
This is an interview featuring me by Dr. Ben Sinclair, a British physician / general practitioner (GP) Sinclair. His podcast is called Health Versus Wealth. Primarily, this podcast is about my health choices and routines. He describes me as a “highly driven entrepreneur, 61 years old (now 62), of THOR Laser company (correction: THOR Photomedicine Ltd and THOR Photomedicine Inc), of which he is is Founder CEO, which specialises in design and manufacture of Photobiomodulation (PBM) devices. THOR £10 million a year in sales, has 5,000 customers in 70 countries, and is marking THOR’s 30th anniversary.” Dr Ben says I am “a businessman who prioritises his wealth, in that he works 364 days a year! (actually, my motivation is establishing PBM as a first-line medical treatment in every department of every hospital in the whole world), wealth might be an outcome, but it is not the goal.) How does this impact his work-life balance? “The only day I … Continue reading
The evidence for Photobiomodulation treatment of musculoskeletal pain
THERAPY EXPO SYNOPSIS: Photobiomodulation (PBM) is one of the best researched physical therapy modalities. More than 800 RCTs have been published on the benefits of PBM, thousands of papers published on the mechanism of action and dose response, more than 8,000 academic papers in total. Systematic reviews and clinical guidelines recommending PBM have been published in the worlds leading journals including The Lancet, British Journal of Sports Medicine, the British Medical Journal and JAMA for a wide range of musculoskeletal disorders. The American College of Physicians recommend PBM for Low Back Pain, and NICE recommend PBM for Oral Mucositis. PBM works primarily on the powerhouse of the cell (mitochondria), increasing energy (ATP) and reducing oxidative stress (cytokines) that lead to inflammation cell death and diseases associated with ageing. The unique feature of PBM is its ability to switch on tissue and regenerative processes. This presentation summarises the evidence for PBM in musculoskeletal pain.
For more on PBM evidence, training or … Continue reading
James Carroll 2020 WALT Presidential Commendation
Thank you to The World Association for PhotobiomoduLation Therapy for the award today. As I said in my acceptance speech I am grateful for the many friends and good times I have enjoyed in the field of LLLT/PBM over the last 33 years, largely thanks to WALT. I should have said more about what I have learned from the many scientists, doctors, dentists, veterinarians, therapists, nurses and patients. It has led to an exciting, rewarding and fulfilling career, and I look forward to the next 40 years as we take PBM to where it is needed and belongs. So thank you WALT for giving me a life worth living.
RIP Dr Kevin Moore who died last week 15 July 2020
 Kevin was a husband, father, anaesthetist, chronic pain specialist, researcher, a fun, kind and generous man.
Kevin was a husband, father, anaesthetist, chronic pain specialist, researcher, a fun, kind and generous man.
He was a vital founding member of the World Association for Laser Therapy (WALT) and a long-standing cheerleader for Photobiomodulation (PBM) as it is now known.
As well as being Consultant Anaesthetist at The Royal Oldham Hospital UK, Kevin was medical director at Dr Kershaw’s Hospice.
Kevin also had the thankless task of being the WALT treasurer for many years. His leadership and steady hand ensured the organisation survived several problematic periods.
All who met him will remember his warmth, humour and generosity of spirit.
He will be forever memorialised in my LLLT/PBM presentations as he researched and published two of my favourite PBM papers of all time (abstracts below and links … Continue reading
LumiThera Enrolls First Patient in the U.S. Multi-Center, LIGHTSITE III Clinical Study to Treat Dry Age-Related Macular Degeneration
SEATTLE, Oct. 1, 2019 /PRNewswire/ — LumiThera Inc., a commercial stage medical device company delivering photobiomodulation (PBM) treatment for ocular disorders and disease, today announced it has begun enrolling patients in a multi-center United States clinical study in dry Age-Related Macular Degeneration (AMD) patients.
The randomized, multi-center study called LIGHTSITE III enrolled and treated the first patient at Cumberland Valley Retina in Hagerstown, Maryland. The United States study is being conducted in leading retinal centers throughout the United States. The study will enroll approximately 100 patients suffering from dry AMD and treat them over the course of two years. In addition to demonstrating safety, key efficacy endpoints include visual acuity, contrast sensitivity and reduction of drusen deposits.
In February, the company announced that the National Institute of Health and the division of the National Eye Institute was providing a $2.5M grant to partially support the U.S. study.
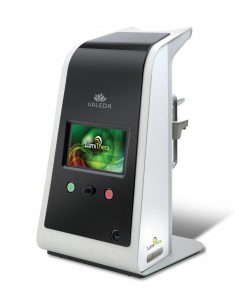
Valeda Light Delivery System Receives 2019 Medical Design Excellence Award
SEATTLE, June 12, 2019 /PRNewswire/ — LumiThera, Inc. and Product Creation Studio today announced that LumiThera’s Valeda Light Delivery System has been honored with a silver award in the 21st Annual Medical Design Excellence Awards competition. The 2019 winners were announced at the MDEA Ceremony on Tuesday, June 11, 2019 in conjunction with the MD&M East event in New York, New York.
LumiThera, Inc. is a leader in the use of photobiomodulation for the treatment of acute and chronic ocular diseases and disorders. Age-related macular degeneration (AMD) is the leading cause of blindness in adults over 65. Product Creation Studio worked with LumiThera from product concept through transfer to manufacturing to design and engineer a medical device that would accurately, safely, and effectively treat patients suffering from dry AMD. The resulting Valeda Light Delivery System is currently the only approved treatment using
Essays
Links
 Featured Testimonials
Featured Testimonials